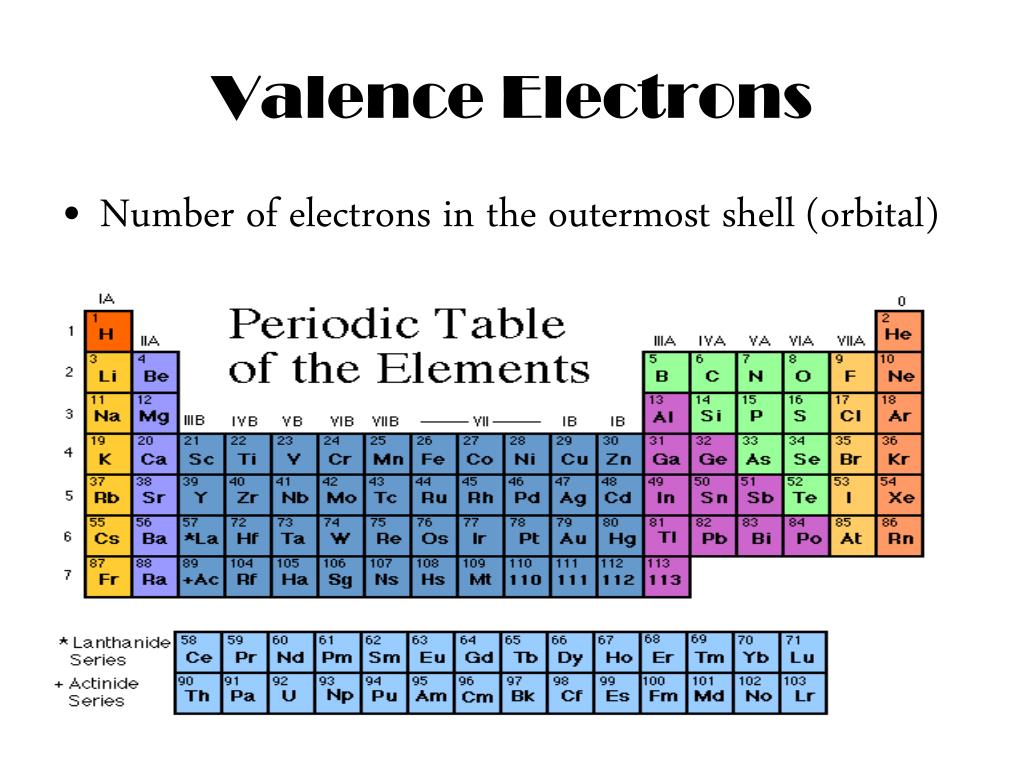
We number the elements, beginning with hydrogen, number one, in integers up to the largest number. As you read down the chart from top to bottom, a line of elements is a Group or Family. As you read across the chart from right to left, a line of elements is a Period. The Periodic Chart of the Elements is just a way to arrange the elements to show a large amount of information and organization. The Periodic Chart of Table of the Elements Overview The Periodic Chart of Table of the Elements.Explanation of States of Matter Problems.SN1SN2 – Nucleophilic Substitution Reactions.

Explanation of Numbers and Math Problems – Set 3.Explanation of Numbers and Math Problems – Set 2.Explanation of Numbers and Math Problems – Set 1.Metric Prefixes and Their Origins and Use.Drawing Cyclohexane Rings – Organic Chemistry.Alkanes and Alkenes – Organic Chemistry.Light & Dark Reactions in Photosynthesis.The valency of the compound is 1 and 1.Search Lessons Search for: Science Resources and Science Lessons – Science Help.Some formulas through which we can make compounds by knowing the valency of their elements are as follows: Hydrogen Chloride Hence, the valency of vanadium are 2,3,4 and 5.įormulas of Compounds by Valence Electrons.Its electronic configuration is Ar 3d 3 4s 2.It is a transition element and most of these show variable valences.It will gain 3 more electrons to complete its octet.Its electronic configuration is 2, 8, 5.It shows two valences: 1 (or Cu(I)), known as cuprous, and 2 (or Cu(II)), known as Cupric.It is a transition element, and most of these elements have variable valences.The outermost shell is already filled, so it will not lose electrons.Sodium will lose 1 electron to complete its octet.Its electronic configuration is 2, 8, 1.Here are the formulas of Valence Electrons for some elements: Sodium Read More: Polytetrafluoroethene (Teflon) The periodic table of all the elements is mentioned below: If there are 8 electrons in the outermost shell, it means that the octet is completed already, and there is no need to lose or gain electrons.If there are 4 electrons in the outermost shell, it can lose or gain 4 electrons to complete its octet.If there are 5, 6, or 7 electrons in the outermost shell, it will gain some electrons to complete its octet.If there are 1, 2, or 3 electrons in the outermost shell, it will lose these electrons to complete its octet.Another way to calculate the valence electron is by determining the electronic configuration of an element.įor Electronic configuration, we have to check the outermost shell of an electron in the following ways:.With the help of group numbers, we can easily calculate the number of valence electrons of an element.Valence electrons can be determined by checking the vertical column of the periodic table.The structure of these elements could be much better. The electrons of these groups can be determined easily, but they are difficult to predict. It can be found in all elements, even if they are from Group 3 to Group 12. Valence electrons can easily be determined with the help of the periodic table.
